
This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. Spinal Column Stimulation (SCS) Systems Intended Use Poster presented at the biannual meeting of the International Neuromodulation Society, London, England. A prospective clinical evaluation of a rechargeable implantable pulse generator (IPG): Final analysis of the sustainability of spinal cord stimulation therapy for chronic lower back pain. Poster presented at: 2018 North American Neuromodulation Society (NANS) Annual Meeting January 11-14, 2018 Las Vegas, NV. Randomized, controlled trial assessing burst stimulation for chronic pain: two-year outcomes from the SUNBURST Study. Treatment-limiting complications of percutaneous spinal cord stimulator implants: a review of eight years of experience from an academic center database. Novel Intermittent Dosing Burst Paradigm in Spinal Cord Stimulation. Deer TR, Patterson DG, Baksh J, et al.Jude Medical’s Prodigy Chronic Pain System with burst technology. De Ridder D, Vanneste S, Plazier M, Vancamp T.Success using neuromodulation with BURST (SUNBURST) study: results from a prospective, randomized controlled trial using a novel burst waveform. Deer T, Slavin KV, Amirdelfan K, et al.Conduct a trial stimulation period before implanting a spinal cord stimulator (SCS)-letter to health care providers. Spinal cord stimulation: clinical efficacy and potential mechanisms. adult population: new data from the 2010 national health interview survey. Prevalence of persistent pain in the U.S. Kennedy J, Roll JM, Schraudner T, Murphy S, McPherson S.Refer to the Instructions for Use for full details on the MR Conditional scan parameters. Refer to the IFU for additional information. 
Safety comparisons and specific dose-response curves for each dosage have not been clinically established. NOTE: In neurostimulation therapy, ‘dose’ refers to the delivery of a quantity of energy to tissue. 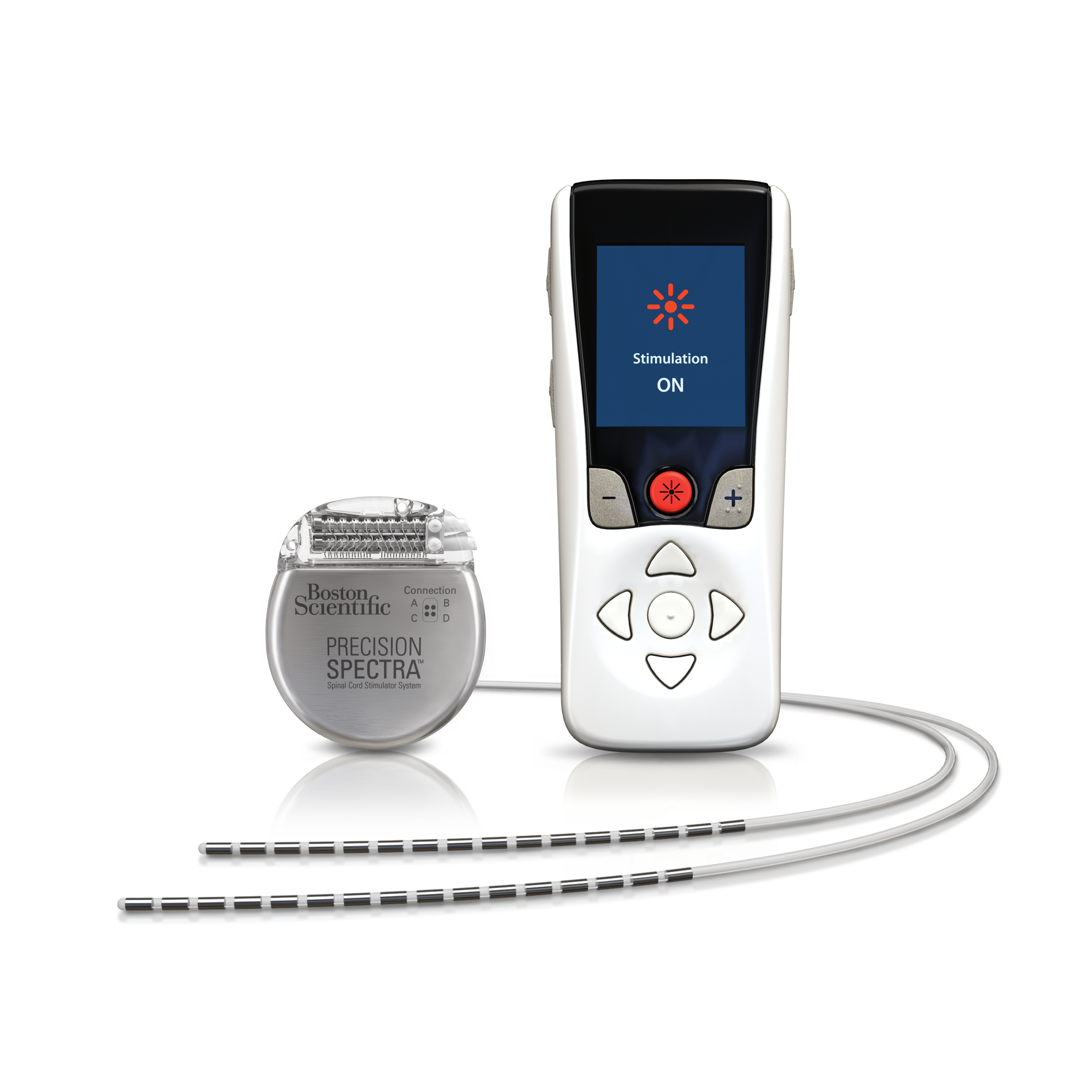
*** Up to 10 years of battery longevity at the lowest dose setting: 0.6mA, 500 Ohms, duty cycle 30s on/360s off. †† Up to 10 years of battery longevity at the lowest dose setting: 0.6mA, 500 Ohms, duty cycle 30s on/360s off. For a list of personal Apple ‡ mobile digital devices compatible with Abbott’s Patient Controller app, visit OR ‡‡ Available on eligible Apple ‡ mobile digital devices. ** Pain and suffering as measured by VAS. † Anywhere with a cellular or Wi-Fi ‡ connection and a sufficiently charged patient controller. * BurstDR™ stimulation superiority when compared to traditional tonic stimulation as studied in SUNBURST ® Bluetooth and Bluetooth logo are registered trademarks of Bluetooth SIG, Inc. ‡ Indicates a third-party trademark, which is property of its respective owner. Be sure to talk to your doctor about the risks associated with the placement of a neurostimulation system. Complications such as infection, swelling, bruising and possibly the loss of strength in or use of an affected limb or muscle group (i.e., paralysis) are possible. Please note: The placement of the leads is a surgical procedure that exposes you to certain risks. If, at the end of the evaluation period, you and your doctor decide the Proclaim™ Plus or Proclaim™ XR SCS System is right for you, you can choose to have the system implanted. Improves your ability to perform daily activities.Your pain management doctor will determine if you are a candidate for stimulation therapy with the Proclaim™ Plus SCS System or Proclaim™ XR SCS System, and may recommend a temporary evaluation.ĭuring the temporary evaluation, you can see whether the therapy: One of the advantages of the Proclaim™ Plus and Proclaim™ XR SCS Systems is that you can try them out to see how well they work for you before committing to an implanted system.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
